- Blog
- Minecraft Metro Hack client
- 64 bit virtualbox on windows 7
- Download 8 ball pool by miniclip
- Youtube free mp3 download music
- Loading icon gif transparent
- Windows 10 clocks on desktop
- Religious vaccine exemption letter pdf
- Grid type micro inverter system
- Position chart trombone
- Expense tracker free printable
- Percy jackson lightning thief full movie
- Grateful dead dancing bear ring
- Honey bee 2 malayalam full movie download
- 1250 sentry safe lost combination
- Strategic group map southwest airlines
- Multiplication worksheets 6 and 7 times tables
- Microsoft office 365 2016 product key free
- Concrete block prices
- Freedom planet playable torque
- Hillbilly rock hillbilly roll chords
- Barbie airplane coloring pages
- Minecraft steve side view
- Interpreting moca test scores
- Best youtube convert to mp3 app
- Download keygen sony vegas pro 13 bagas31
- Omni volume calculator triangular prism
- Tasks by planner and to do teams
- Play scrabble against the computer online free
- Gacha life free game play on chromebook
- Sphax texture pack 1-7-10 modded
- Specific density of water
- Coreldraw graphics suite x7 full version
- Us mail forward service
- Circle linkedin png logo
- Anime live wallpapers pc
- Business Intelligence Development Studio sql server 2012
- Christian wedding ceremony script for emcee
- Diet plan ecto mesomorph
- Nhd website rough draft example
- Fill ds 160 form
- Timeworks clocks parts
- Mixed in key 8 torrent mac

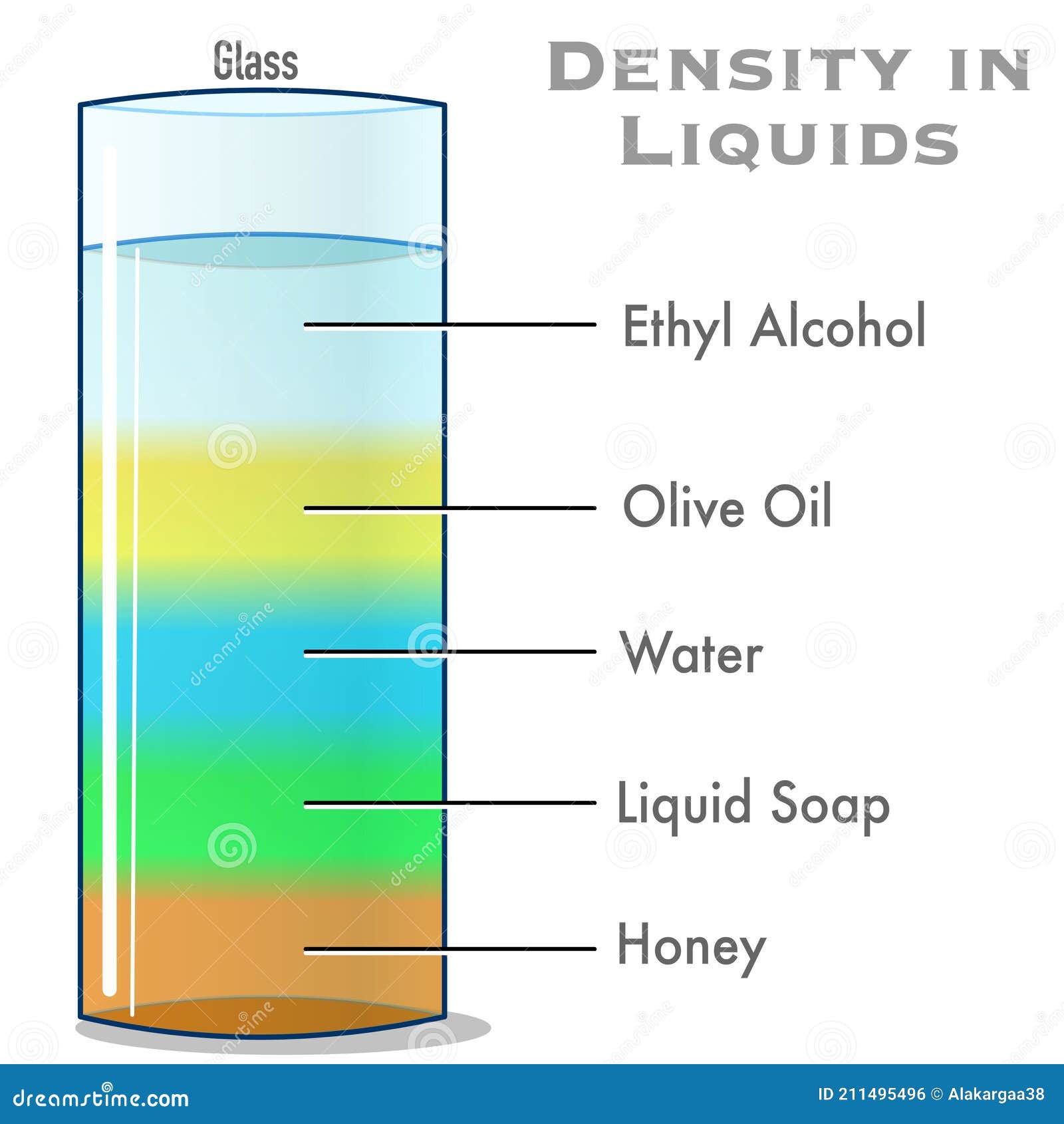
The higher value (HHV), or gross heat of combustion, includes all the heat released as the products cool to room temperature and whatever water vapor is present condenses.There are two kinds of heat of combustion: But as a source of heat or for use in a heat engine, the relevant quantity is the change in standard enthalpy or the heat of combustion. This is given by the change in standard Gibbs free energy. Another is the theoretical amount of electrical energy that can be derived from reactants that are at room temperature and atmospheric pressure. One is the theoretical total amount of thermodynamic work that can be derived from a system, at a given temperature and pressure imposed by the surroundings. There are several different types of energy content. Electrochemical reactions are used by most mobile devices such as laptop computers and mobile phones to release energy from batteries. Liquid hydrocarbons (fuels such as gasoline, diesel and kerosene) are today the densest way known to economically store and transport chemical energy at a large scale (1 kg of diesel fuel burns with the oxygen contained in ≈15 kg of air). Chemical reactions are used by organisms to derive energy from food and by automobiles to derive energy from gasoline. Nuclear reactions take place in stars and nuclear power plants, both of which derive energy from the binding energy of nuclei. In order of the typical magnitude of the energy released, these types of reactions are: nuclear, chemical, electrochemical, and electrical. There are different types of energy stored in materials, and it takes a particular type of reaction to release each type of energy. A pressure gradient describes the potential to perform work on the surroundings by converting internal energy to work until equilibrium is reached. Likewise, the energy required to compress a gas to a certain volume may be determined by multiplying the difference between the gas pressure and the external pressure by the change in volume. For example, the energy density of a magnetic field may be expressed as and behaves like a physical pressure. In cosmological and other general relativistic contexts, however, the energy densities considered are those that correspond to the elements of the stress-energy tensor and therefore do include mass energy as well as energy densities associated with pressure.Įnergy per unit volume has the same physical units as pressure and in many situations is synonymous. Often only the useful or extractable energy is measured, which is to say that inaccessible energy (such as rest mass energy) is ignored. It is sometimes confused with energy per unit mass which is properly called specific energy or gravimetric energy density. In physics, energy density is the amount of energy stored in a given system or region of space per unit volume.
- Blog
- Minecraft Metro Hack client
- 64 bit virtualbox on windows 7
- Download 8 ball pool by miniclip
- Youtube free mp3 download music
- Loading icon gif transparent
- Windows 10 clocks on desktop
- Religious vaccine exemption letter pdf
- Grid type micro inverter system
- Position chart trombone
- Expense tracker free printable
- Percy jackson lightning thief full movie
- Grateful dead dancing bear ring
- Honey bee 2 malayalam full movie download
- 1250 sentry safe lost combination
- Strategic group map southwest airlines
- Multiplication worksheets 6 and 7 times tables
- Microsoft office 365 2016 product key free
- Concrete block prices
- Freedom planet playable torque
- Hillbilly rock hillbilly roll chords
- Barbie airplane coloring pages
- Minecraft steve side view
- Interpreting moca test scores
- Best youtube convert to mp3 app
- Download keygen sony vegas pro 13 bagas31
- Omni volume calculator triangular prism
- Tasks by planner and to do teams
- Play scrabble against the computer online free
- Gacha life free game play on chromebook
- Sphax texture pack 1-7-10 modded
- Specific density of water
- Coreldraw graphics suite x7 full version
- Us mail forward service
- Circle linkedin png logo
- Anime live wallpapers pc
- Business Intelligence Development Studio sql server 2012
- Christian wedding ceremony script for emcee
- Diet plan ecto mesomorph
- Nhd website rough draft example
- Fill ds 160 form
- Timeworks clocks parts
- Mixed in key 8 torrent mac
